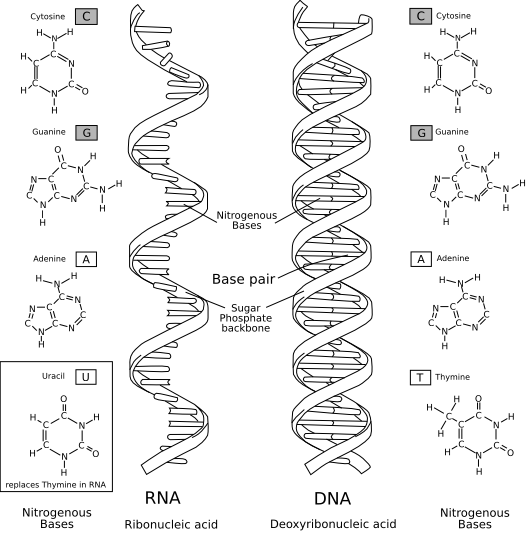
A nucleic acid is a polymer of subunits called nucleotides, each of which contains a five-carbon sugar molecule, a phosphate ion, and a nucleobase. In a given nucleic acid, the only differing characteristic of nucleotides is their bases, meaning that a molecule of nucleic acid can be identified by its sequence of bases. The only two naturally appearing nucleic acids are RNA (whose sugar is called ribose) and DNA (whose sugar is deoxyribose). DNA and RNA both have four possible nucleobases, three of which are common to both nucleic acids; thymine in DNA is replaced with uracil in RNA.
Nucleic acids, discovered by Friedrich Miescher in 1869, are common to all life forms found on Earth, including life-like structures such as viruses. The reason for their universality is that nucleic acids (and particularly DNA) are responsible for encoding traits: DNA is transcribed into RNA, which is translated into proteins that drive cellular functions, which eventually manifests itself as traits in a multicellular organism.
The figure below illustrates the structure of RNA and DNA. Both nucleic acids are formed by sugar and phosphate molecules
linking together into an alternating chain called a sugar-phosphate backbone. Specifically, the five carbon atoms
of the sugar molecule are arranged in a clockwise ring and labeled 1', 2', 3', 4', 5'. The 5' carbon
atom of one sugar molecule is then linked to the phosphate ion (
In contrast to single-stranded RNA, the bases of a strand of DNA bond with bases in an opposing strand to form a double-stranded molecule (adenine bonds with thymine and cytosine bonds with guanine), which then twists into a double helix shape.